How to clean and passivate large stainless steel surfaces
Weld cleaning | Stainless steel | Thursday, 22 February 2018
How to clean large stainless steel structures?
Stainless steel and to a lesser degree, aluminum, are among the most valued metals. Both have qualities of durability and resistance to corrosion. Unfortunately, no metal is perfect. Stainless steel and aluminium are susceptible to corrosion. This is why proper cleaning and passivation is important when these metals have exposure to environments where corrosion is possible. Before explaining options for cleaning and passivating large metal (stainless steel and aluminum) surfaces, it is important to understand how corrosion forms.

Stainless steel is a unique metal. It actually forms its own layer of iron-chromium oxide as a deterrent against corrosion. This layer protects the metal. However, harsh chemicals, salt-laden environments such as constant exposure to saltwater, and damage to the surface by mechanical action such as cutting will create openings in the layer. Welding also breaks down the protective layer.
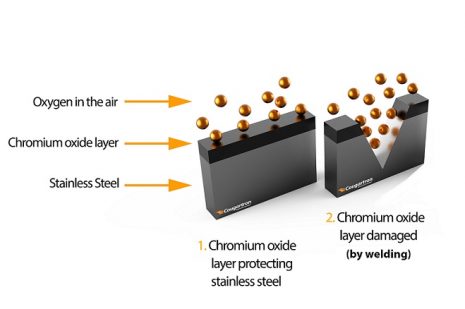
Stainless steel, unlike other metals, is unique in that it attempts to repair the surface naturally through a chemical interaction between oxygen in the environment and the stainless steel. In a perfect world, this natural reaction would form a new bond with the same protection. Because some environments are filled with contaminants, the result is far from perfect. These contaminants settle in areas where the original iron-chromium oxide was disrupted.
The contaminants prevent the complete interaction of the oxygen and the stainless steel. Instead of forming a corrosion resistant barrier, the barrier formed traps the contaminants creating the potential for corrosion.
The cost of repairing damage from corrosion is extremely expensive. The United States Department of Defense estimates it spends in excess of $22 billion every year for the repair, maintenance, and prevention of corrosion. This is why governments and companies dedicate so much effort to finding methods to clean and maintain metals including stainless steel and aluminum. Proper cleaning plays an important role in metal (stainless steel) maintenance.
Cleaning and passivation options
There are several options when it comes to passivation. Ultrasonic, pickling, and electrochemical processes all remove corrosive contaminants, allowing the restoration of the protective layer to stainless steel. However, not all have the capability or practicality when it comes to cleaning and the passivation of large surfaces.
Here are the two most common processes:
- Chemical pickling
- Electrochemical cleaning
Chemical Pickling
Chemical pickling, or pickling for short, has a long history of use for cleaning corrosion from stainless steel. The process is somewhat flexible. A chemical pickling agent made from nitric acid and hydrofluoric acid is applied to a surface to remove contaminants. The hydrofluoric acid removes the contaminants while the nitric acid aids in activating the stainless steel surface to promote passivation.
The application of the pickling agent varies depending on the size of the surface. Common methods include brushing or spraying; however, dipping larger parts or surfaces in a large bath filled with the pickling solution is used in some applications. The benefits of dipping allow the pickling solution to reach everywhere on the surface and covering hard to reach areas.
Pickling as a means of cleaning and passivation does come with significant hazards.
Electrochemical method
As the name implies, an electrochemical process relies on both an electrical charge and a chemical reaction. This process also provides effective cleaning and passivation of large surfaces, but with significant differences compared to pickling. The difference is in terms of safety and speed (efficiency).
The electrochemical process also uses an acid (electrolyte) to clean and provide passivation. However, the acid used in electrochemical cleaning is phosphoric acid, an ingredient used in soda pop. This does not mean you do not have to take precautions. But the degree of precautions is far less than working with the pickling process.

When cleaning larger sheet metal and steel structures, the benefits of the electrochemical process are quite evident.
- Apart from speed and safety, work flexibility is another important factor to consider. Electrochemical weld cleaning is not a stationary process – it can be done on- and off-site, depending on your needs.
- All of this without extensive protective equipment and complex work protocol prescribed for chemical pickling.
Since regulations for dealing with the pickling paste are rigorous, companies often have to outsource the work to other firms that possess the necessary capacity and work standards. In addition to extra cost, this also has a negative impact on lead times and supply chain flexibility. Moreover, the pickling paste is quite ineffective under low temperatures so it cannot be used outside during cold weather – another area in which electrochemical weld cleaning is a more favorable option.
Despite the obvious shortcomings, pickling was often preferred when cleaning larger surfaces since it provided faster results than other methods.
Electrochemical weld cleaning systems eliminate most of these issues. As they are essentially harmless and easy to operate, they can be used immediately after welding anyplace they are needed.
Our most powerful models such as Cougartron FURY 100 and FURY 200 are powerful enough to successfully and quickly eliminate all the impurities from stainless steel – even on extremely long welds and large assemblies (tanks).

The chemical electrolyte solution in this process is activated with a mild electric charge to remove corrosion and promote passivation. Recent improvement in electrochemical technology now makes it possible to clean polish and passivate large surfaces in one step; and do so quickly.
This new technology makes it possible to use on a variety of welds including Tig, Mig, MMA, Stick, and other types of welds. It also works on all stainless steel grades and aluminium.
Contact us to find the best solution for your needs
You can find out more about the benefits of this new electrochemical cleaning and passivation technology by contacting us today. Our dedicated team will work with you to find the best solution for your production requirements. We can arrange a machine demonstration at your location with one of Cougartron’s representatives or partners all over the globe.
Loading...
Please wait while the form loads.
Most Powerful Weld Cleaning Machines
Loading products...
Please wait while the products load.
Cougartron Weld Cleaning Fluids
Loading products...
Please wait while the products load.


 English
English  English (US)
English (US)  German
German  Danish
Danish  Swedish
Swedish  French
French  Polish
Polish  Spanish
Spanish 

