Ask Cougartron Part 2: Passivation of stainless steel after welding
Weld cleaning | Ask Cougartron | Stainless steel | Thursday, 15 February 2018
Stainless Steel Passivation after Welding
It’s time to answer another question we received through Ask Cougartron – our new monthly Q/A feature.
YOUR QUESTION: With stainless steel being naturally corrosion-resistant, why does it have to be passivated after welding?
Before answering this question, it is important to first understand the composition and physical properties of stainless steel.
So, let’s begin.
Is stainless steel naturally passive?
In its essence, stainless steel is a steel alloy composed mainly of iron with the addition of chromium (minimum of 10-12%).
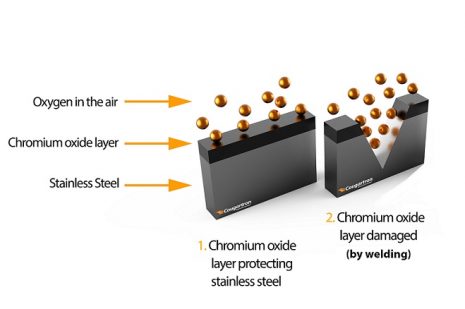
When exposed to normal atmospheric conditions (air), chromium will react with oxygen and create a protective layer on the surface of the alloy.
The passive layer is very thin but strong enough to prevent the penetration of additional oxygen and moisture into the interior. This is the reason why stainless steel possesses unparalleled anti-corrosion properties.
So, is stainless steel naturally passive? The answer is yes.
Can stainless steel rust?
The stainless steel surface becomes active when the chromium oxide layer is damaged or broken.
If the stainless steel surface is not significantly damaged, the passive layer will likely restore itself naturally when a new reaction between chromium and oxygen particles occurs.
However, various industrial processes such as cutting, grinding, and welding can irreversibly destroy the protective layer on the steel surface.
This creates ideal conditions for the formation of rust and other types of contamination.
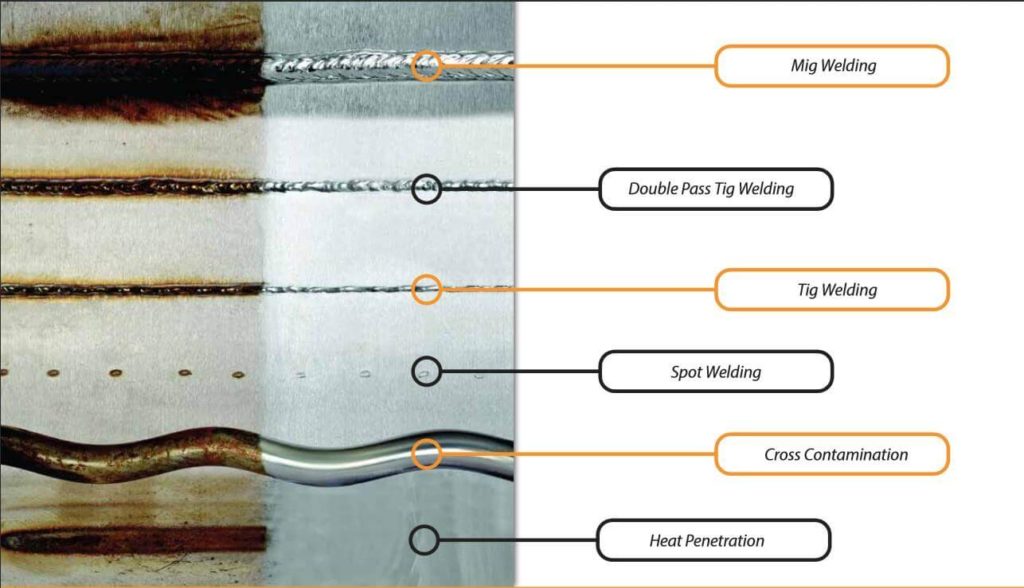
Loss of passive layer after welding
Pressure and heat as integral parts of the welding process are directly responsible for the formation of rust on stainless steel surfaces.
As the steel structure changes through melting, the free iron particles begin to accumulate on the surface where they react with oxygen and cause corrosion.
As explained earlier, the stainless steel would not be stainless without a protective chrome layer.
The concentration of chromium particles decreases even in the heat-affected zones – not just on the surface exposed to melting.
When this occurs, the passive state of the stainless steel has to be artificially restored.
However, effective passivation is impossible without a thorough post-welding cleaning and treatment of the surface.
The protective chromium-oxide layer cannot fully re-emerge unless all contaminants are removed from the affected areas.
Why grinding does not guarantee weld passivation?
Welding often results in a significant number of imperfections and flaws appearing on stainless steel surfaces. Some of the distinguished examples are rust, discoloration, burn marks, and heat tints.
Traditional methods like mechanical grinding or wire brushing can be quite effective when dealing with these issues. However, the effect of this process is mostly aesthetic in nature.
The mechanical removal of contaminating particles is not restoring the passive layer on the stainless steel surface. It can provide a solid prerequisite for this to occur naturally but there is no guarantee that the chromium oxide layer will form fast enough to prevent the reappearance of rust.
Improper use of mechanical grinding machines can also destroy the surface completely and stop the formation of a new passive layer.
Additionally, cleaning welds with abrasives can lead to more unwanted consequences like the altered appearance of the surface and scratches.
So, how do we properly clean and protect the stainless steel surface?
Electrochemical process – cleaning and passivation in one pass
The combination of electricity and specialized electrolytic fluids is sufficient for successful cleaning and passivation of the surface – in one step.
The electrochemical process will not only remove imperfections on the surface after welding – but it will also restore the oxide layer that returns stainless steel back to its normal passive state.
Unlike grinding, this cleaning method will successfully remove all dirt and discoloration from the welds. This is especially important for the successful restoration of the passive layer.
Cougartron weld cleaning machines are designed to initiate and induce the formation of chromium oxide film on stainless steel surfaces and welds. Thus, you can be sure that rust and other contaminating factors will not reappear after they are successfully eliminated.

Cleaning and passivation of the heat-affected zone
The heat-affected zone (HAZ) is an area on the metal surface whose microstructure has been altered under the influence of heat during welding.
It’s a non-melted portion of the metal surface found between the unaffected base metal and the weld metal (see image below).

The size of the heat-affected zone depends on several factors
- amount of heat
- fabrication method
- type of material used
However, its negative consequences are consistent and always present.
The change in the microstructure makes the steel weaker and less resistant to corrosion and stress cracking.
Proper post-weld treatment (cleaning and passivation) is the only way to recover the structural stability of steel and prevent further decay.
But to what extent? How much of the surface do you actually need to treat? The entire metal assembly or just the heat-affected zone (and the weld)?
The answer is actually very simple.
The best approach is to actively clean and passivate only the part of the surface that was actually affected by the welding process. This in all cases includes the actual weld and the heat-affected zone (areas where discoloration and other post-weld imperfections usually appear).
The application of weld cleaning fluids on unaffected areas will not damage them. In any case, you will always reach parts of the metal surface outside the heat-affected zone. However, the point is that these areas do not require focused and active treatment.
Side effects of electrochemical method
Electrochemical cleaning is done by using a food-grade phosphorus-based acid.
Although mild, acids often leave a white residue on the surface of the steel after cleaning and passivation.
Removal of these white acid marks is done for two main reasons – lowering the pH value and preserving the attractive appearance of the surface.
Since the residue can sometimes become visible only after a couple of days, it is important to neutralize the surface immediately or soon after cleaning and passivation.
In most cases, it is impossible to solve this problem with water unless a pressure washer is utilized. This is why specialized neutralizing fluids have to be used. We recommend using our CGT-N5 or contacting us for advice in case of persistent issues.
How can you succesfully passivate your stainless steel?
Help us understand your concerns and challenges better – together we can improve the general quality of weld cleaning in the industry. Fill out the form below to ask a question, get a free consultation or a Cougartron machine demo.
Loading...
Please wait while the form loads.


 English
English  English (US)
English (US)  German
German  Danish
Danish  Swedish
Swedish  French
French  Polish
Polish  Spanish
Spanish 

